Abstract
Introduction: Myelodysplastic syndromes (MDS) comprise a group of clonal hematopoietic disorders characterized by ineffective hematopoiesis, cytopenias, dysplasia in ≥ 1 cell line, cytogenetic and molecular abnormalities, and variable risk of progression to acute myeloid leukemia. Treatment goals for MDS classified as lower-risk (LR-MDS) include transfusion independence, improvement in hemoglobin (Hb) levels, and maintenance of or improvement in quality of life. Erythropoiesis-stimulating agents (ESAs) are the first-line (1L) treatment for anemia in most patients with LR-MDS lacking del(5q), but a proportion of patients do not respond to ESA treatment, or experience progression of anemia despite ESA treatment. The objective of this real-world analysis was to describe patient characteristics, treatment patterns (including ESA use), and outcomes in patients with LR-MDS.
Methods: A retrospective, observational, US multisite, cohort study was conducted among adult patients initially diagnosed with LR-MDS between January 1, 2017 and June 25, 2020. Eligible patients had ≥ 1 year of follow-up after diagnosis (unless the patient died during this time) and did not receive luspatercept or any MDS treatment as part of a randomized, controlled trial. Community oncologists abstracted data from medical records, and descriptive statistics were used to summarize patient characteristics, treatment patterns, and outcomes. Data presented are from an interim analysis of an ongoing study; the last date of data collection for this analysis was July 6, 2021.
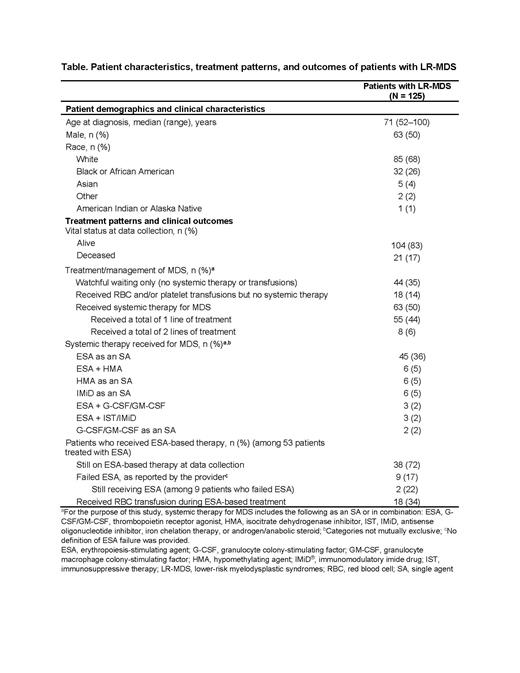
Results: Among 125 eligible patients with LR-MDS, median follow-up time was 16 months, and 83% of patients were still alive at the time of data collection. In the 8 weeks prior to diagnosis, 54% of patients did not receive any red blood cell (RBC) or platelet transfusions, while 42% and 4% had low or moderate transfusion burden, respectively.
Overall, 75% of patients were negative for del(5q), and 80% of patients were negative for ring sideroblasts (RS). At diagnosis of LR-MDS, serum erythropoietin levels were < 200 U/L in 49% of patients, ≥ 200 U/L in 25%, and unknown in 26%.
In terms of disease management, 35% of patients did not receive any systemic therapy or transfusions, 14% received RBC and/or platelet transfusions, but no systemic therapy for MDS, and 50% were reported to have received ≤ 2 lines of systemic therapy following LR-MDS diagnosis (Table). Of the 42% of patients who received an ESA, 70% received an ESA for MDS as a single agent (SA) only, 15% as combination therapy only, and 8% as both SA and combination therapy. A further 8% received SA ESA followed by a non-ESA-based regimen. Among 45 patients who received SA ESA as 1L treatment, 18% went on to receive a hypomethylating agent (HMA) or immunomodulatory imide drug as an SA, or ESA combined with HMA or a granulocyte or granulocyte-macrophage colony-stimulating factor (G-CSF/GM-CSF) in a subsequent line. Among patients who received an ESA, 72% were still on ESA-based therapy at the time of data collection.
Abstracting physicians reported that 17% of patients who received an ESA failed ESA treatment. Among these patients, physicians' determination of ESA failure was based on the National Comprehensive Cancer Network guidelines/International Working Group 2006 response criteria for 78% of patients, and on physicians' own clinical judgement for 22%. At data collection, ESA was still being administered to 22% of patients considered to have failed ESA treatment. Finally, 34% of those who received an ESA also received RBC transfusion(s) during ESA-based treatment and therefore were not transfusion independent for the entire duration of ESA-based treatment (Table).
Conclusions: Results from this real-world cohort study indicate that over a third of patients with LR-MDS have been managed using watchful waiting only, with no systemic treatment or transfusions received; among those patients who received treatment, most received an ESA. Nearly one-fifth of patients in this study treated with an ESA were considered to have failed ESA treatment by abstracting physicians, though this proportion is likely to be higher with extended follow-up; 2 of 9 patients who failed ESA treatment were still receiving an ESA. Further research including longer follow-up is warranted to understand how patients with LR-MDS respond to different treatment regimens.
Zimmerman Savill: Cardinal Health: Current Employment; Roche/Genentech: Ended employment in the past 24 months. Gajra: Cardinal Health: Current Employment, Current equity holder in publicly-traded company. Price: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Kish: Cardinal Health: Current Employment, Current equity holder in publicly-traded company, Research Funding. Brown-Bickerstaff: Cardinal Health: Current Employment. Falkenstein: Cardinal Health: Current Employment. Miller: Cardinal Health: Current Employment. Laney: Cardinal Health: Current Employment. Mukherjee: Acceleron: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Research/Independent Contractor, Research Funding; McGraw Hill: Honoraria, Other: Editor of Hematology Oncology Board Review (ongoing); Celgene Corporation: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Research/Independent Contractor, Research Funding; AbbVie: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees; AAMDS in Joint Partnership with Cleveland Clinic Taussig Cancer Institute: Honoraria; Eusa Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Teaching and Speaking; Jazz Pharmaceuticals: Research Funding; Bristol-Myers Squibb Co.: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; BioPharm: Consultancy; Partnership for Health Analytic Research: Honoraria; Blueprint Medicines: Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal